Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Arai, Shigeki; Yonezawa, Yasushi*; Tamada, Taro; Tokunaga, Hiroko*; Ishibashi, Matsujiro*; Tokunaga, Masao*; Kuroki, Ryota
no journal, ,
no abstracts in English
Adachi, Motoyasu; Shimizu, Rumi; Kuroki, Ryota; Moriya, Keisuke*; Kidokoro, Shunichi*; Hidaka, Koshi*; Tsuda, Yuko*; Kiso, Yoshiaki*
no journal, ,
Human immune deficiency virus protease-I (HIV-PR) is one of the important drug target proteins for the acquired immune deficiency syndrome. In this study, we designed the two single chain derivatives of wild-type and A17 type HIV-PRs in which the catalytic residue of Asp25 was placed with Asn25 to inactivate the enzyme. The tertiary structure of sc-HIV-PR of wild-type and A17 type were determined by X-ray crystallography to 1.1 and 1.5  resolution, respectively. The both complex structures showed that Asn25 forms hydrogen bond with carbonyl group of inhibitor.
resolution, respectively. The both complex structures showed that Asn25 forms hydrogen bond with carbonyl group of inhibitor.
Shimizu, Rumi; Adachi, Motoyasu; Kuroki, Ryota; Blaber, M.
no journal, ,
We have already succeeded in creation of the de novo designed protein (Symfoil) exhibiting the threefold symmetrical  -trefoil fold based on the human acidic fibroblast growth factor. Based on the Symfoil protein, we created Symfoil-II having the three repeats. The Symfoil-II protein was expressed in Eschericha coli as soluble protein, and purified by metal affinity chromatography using nickel-chelated agarose. The Symfoil-II was further purified by anion-exchange column chromatography after removing the HisTag by proteolysis. Symfoil-II was crystallized in 0.1 M Tris-HCl buffer (pH7.0) containing 1.8 M ammonium sulfate as precipitant at 20
-trefoil fold based on the human acidic fibroblast growth factor. Based on the Symfoil protein, we created Symfoil-II having the three repeats. The Symfoil-II protein was expressed in Eschericha coli as soluble protein, and purified by metal affinity chromatography using nickel-chelated agarose. The Symfoil-II was further purified by anion-exchange column chromatography after removing the HisTag by proteolysis. Symfoil-II was crystallized in 0.1 M Tris-HCl buffer (pH7.0) containing 1.8 M ammonium sulfate as precipitant at 20 C. The X-ray diffraction data was collected to 1.9
C. The X-ray diffraction data was collected to 1.9  resolution using a Rigaku R-AXIS VII diffractometer. The crystal belongs to a monoclinic space group C2. The refined crystal structure of Symfoil-II showed three fold symmetry as is observed in other Symfoils.
resolution using a Rigaku R-AXIS VII diffractometer. The crystal belongs to a monoclinic space group C2. The refined crystal structure of Symfoil-II showed three fold symmetry as is observed in other Symfoils.
 -endoglucanase from
-endoglucanase from 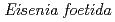
Arimori, Takao*; Ito, Akihiro*; Nakazawa, Masami*; Ueda, Mitsuhiro*; Tamada, Taro
no journal, ,
no abstracts in English
Kosaka, Megumi*; Kanao, Tadayoshi*; Nakayama, Hisayuki*; Yoshida, Kyoya*; Kamimura, Kazuo*; Takada, Jun*; Yamada, Hidenori; Tamada, Taro; Okazaki, Nobuo*; Kuroki, Ryota
no journal, ,
no abstracts in English
Kuroki, Ryota
no journal, ,
Elucidation of the complicated structure of protein molecule will contribute not only to understand molecular mechanism occurring in life events, but also to improve the molecular function of the proteins from industrial point of view. Although neutron crystallography is not common tool to determine the tertiary structure of protein, there are several neutron diffractometers have been constructed for the purpose of protein structure determination. Neutron crystallography is expected to become an important tool used in the field of protein science. In this meeting, I wish to introduce the current status and the future prospects of neutron diffractometer focused on protein structure determination.